Atopic dermatitis (AD) is the most common type of eczema, occurring in up to 3% of the adult population worldwide, and it is the most common skin disease among children, with the disease commonly occurring by five years of age.
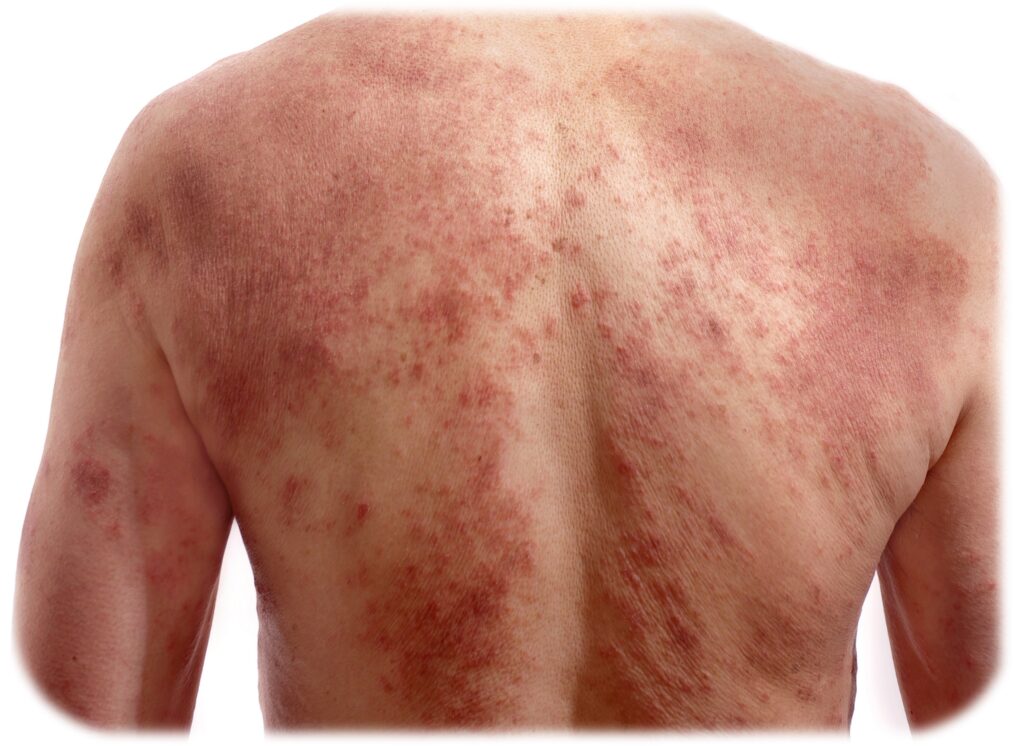
Atopic dermatitis (AD) is characterized by a defective skin barrier, which allows allergens and other irritants to enter the skin, leading to an immune response and inflammation driven by inflammatory cytokines (e.g., IL4 and IL13). This reaction produces a red, itchy rash, most frequently occurring on the face, arms and legs, but it can cover most areas of the body.
Source: Diagnosed patients incl. US, EU5, and Japan (Evaluate Pharma 2023)
Itch is the most problematic symptom
Itch is the most problematic symptom for AD patients. When suffering from itch, patients also frequently suffer from sleep disturbances and emotional problems. Generally, patient with AD experience that the disease has a significant impact on their quality of life and overall health. For example, adults with AD exhibit a significantly increased risk of ADHD, anxiety, depression and suicidal ideation compared to the general population.
Meet Amalie, a 26-year-old woman diagnosed with severe atopic dermatitis.
Meet Emma, a young woman living with atopic dermatitis.
Treatment of AD
Patients with AD are diagnosed based on the severity of the disease (mild, moderate or severe). Generally, patients with AD are treated with topical or systemic (oral or injectable) treatments. Topicals are mostly used by patients with mild AD or used concomitantly with systemic therapies for patients with severe AD. The injectable biological treatments approved for the treatment of AD have had a substantial positive impact for patients. However, many patients using these treatments do not experience 17 satisfactory responses, indicating that more mechanisms of action are needed. The first approved oral therapies, Janus kinase inhibitors (JAKs), are typically the treatment of last resort due to safety concerns and are mainly used for patients with severe AD.
Patient preference for oral treatments
Patients with AD report a strong preference for safety, oral administration and rapid itch relief. None of the current treatment options can deliver on all three parameters. Injectable biologics offer slow relief from itching, and many patients face an injection barrier due to a dislike of needles. The JAKs have significant safety concerns and are limited to non-responders on biologics. The current treatment landscape for AD leaves significant unmet medical need for oral systemic treatments as well as new mechanisms of action. Orismilast holds the potential to become a first-in-class safe oral treatment, and a safe and effective alternative to JAKs and injectable biologics.
Read more about orismilast here


Meet Irene and learn about her experience with atopic dermatitis (eczema). A disease she was diagnosed with when she was 5 years old. Read Irene’s story.
” At times, I can be very frustrated about having atopic dermatitis e.g., when the itching is severe, and the skin is frayed and one cannot live life as all other people”.

